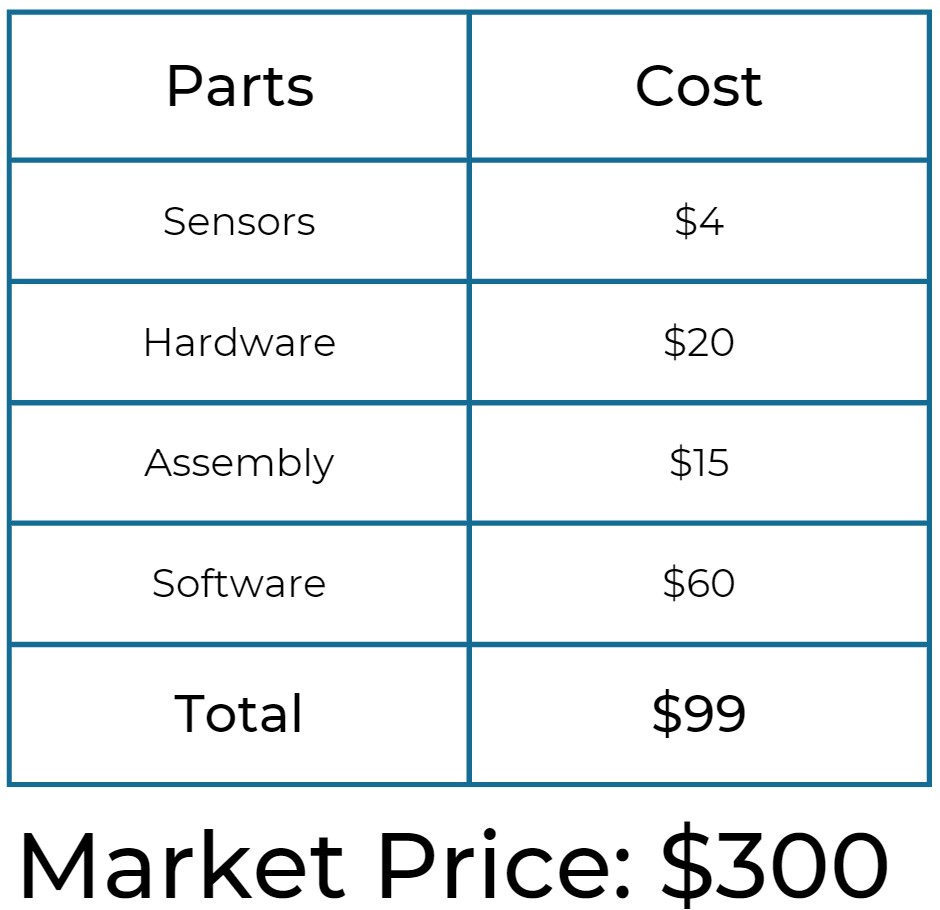
The Product
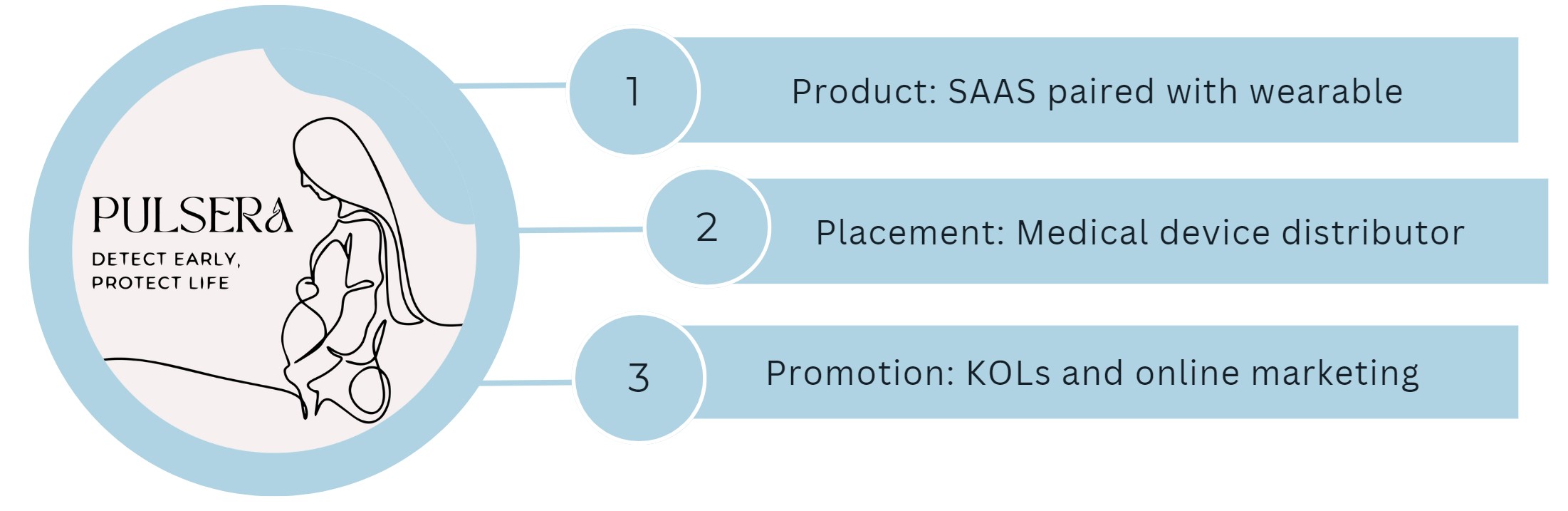
Pulsera offers a software as a service paired with a wearable device. We will partner with medical device distributors to get these into clinics where they can be prescribed to patients. Additionally, the devices will be promoted through key opinion leaders and online marketing. Insurance can help with the cost of the device through CPT codes.
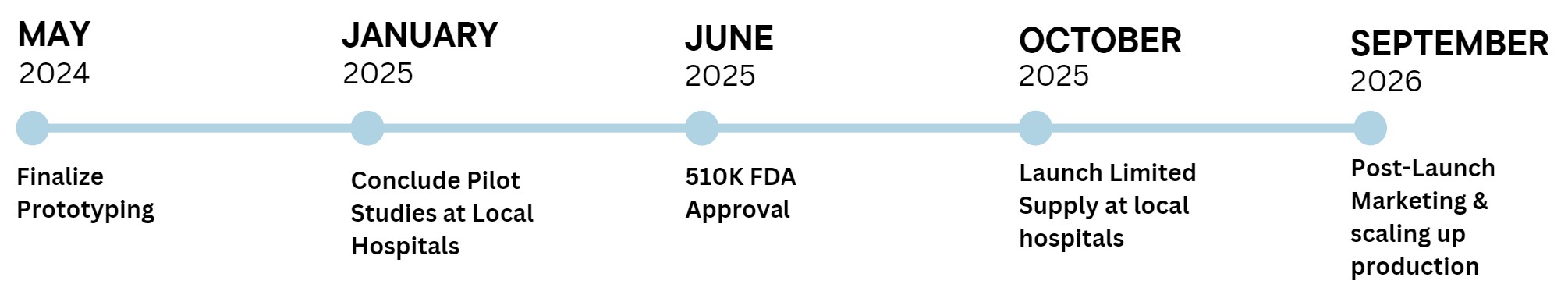
The goal is to finalize the prototype by May 2024 to begin pilot studies at local hospitals. The information and data used from the pilot studies can be used when filing for FDA approval, which will hopefully be approved by June of 2025. In October, we will launch limited supplies to local hospitals in Orange and Los Angeles County. From there, we will do a post-launch marketing and scale up the production of the device.